|
If you get a different value, please let me know. Answerĭiscussion: This number has not been checked. Lattice Energy (kJ mol-1) Experimental Hard-Sphere Born-Landé Born-Meyer Kapustinskii -772 -858 -762 -752 -753 The hard-sphere, Born-Landé and Born-Meyer equations unfortunately require knowledge of the Madelung constant and hence the crystal structure. Evaluate the energy of crystallization, E cryst. Comparison of the experimental and calculated lattice energy for NaCl. Example 1: Compute the Lattice energy of NaCl by using Born-Lande equation. Following this convention, the lattice energy of NaCl would be +786 kJ/mol. Skill: Evaluate the lattice energy and know what values are needed.Īssume the interionic distance for NaCl 2 to be the same as those of NaCl ( r = 282 pm), and assume the structure to be of the fluorite type ( M = 2.512). U L equilibrium value of the lattice energy. AnswerĮnergy of crystallization is -3527 kJ/mol Calculate lattice energy of NaCl, from following data using Born-Habers cycle. Evaluate the energy of crystallization, E cryst for CaO. Lime, CaO, is know to have the same structure as NaCl and the edge length of the unit cell for CaO is 481 pm. Which one of the following has the largest lattice energy? LiCl, NaCl, CaCl 2, Al 2O 3 AnswerĬorrundum Al 2O 3 has some covalent character in the solid as well as the higher charge of the ions. It defines the enthalpy change in a crystal lattice when the structure is formed or broken. 
Skill: Explain the trend of lattice energy. It is calculated by measuring the energy required to break the bonds in the compound and separate the ions, and then subtracting this value from the energy required to form the bonds in the compound. In both of the compounds, Na+ ion is commmon. Which one of the following has the largest lattice energy? LiF, NaF, CaF 2, AlF 3 Answer Lattice enthalpy of ionic compound is inversely proportional to sum of the ionic radius of cation and anion. 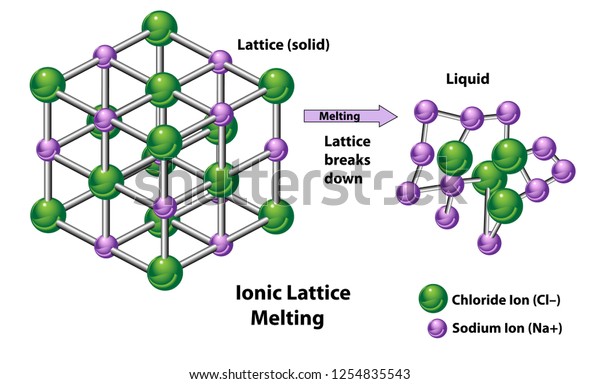
When methods to evaluate the energy of crystallization or lattice energy lead to reliable values, these values can be used in the Born-Haber cycle to evaluate other chemical properties, for example the electron affinity, which is really difficult to determine directly by experiment. 10(-10) m According to Born lange.s equation, lattice enthalpy, Delta('lattice') H 1.39 xx 105 (Z+. Much more should be considered in order to evaluate the lattice energy accurately, but the above calculation leads you to a good start.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
